Technologies
FDA, Pfizer delay authorization for COVID vaccine for kids under 5: What to know
The FDA will review data on all three doses of the vaccine for young kids, instead of starting with just two. The data should be available in early April.
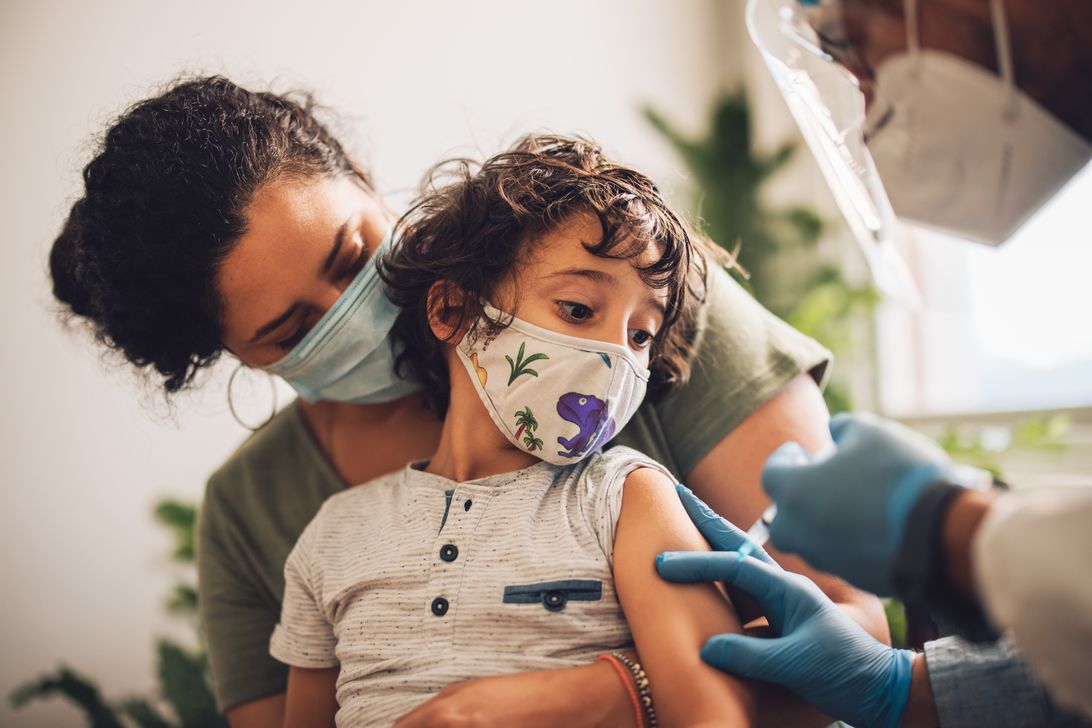
Parents of children under 5 will have to wait a little longer to get their kids vaccinated against COVID-19.
Pfizer and BioNTech announced Friday that the companies are extending their «rolling submission» process for the vaccine for kids 6 months through 4 years and are waiting for more data on all three doses before asking the US Food and Drug Administration to authorize the first two shots. Just two doses of the vaccine weren’t effective for the whole age group.
The FDA said on Friday that the agency is postponing its Feb. 15 committee meeting, which would’ve included a vote on whether to move forward with the first doses of the COVID-19 vaccine for young kids. The agency said that it will provide an updated time for a new meeting once the FDA receives additional data for the full vaccine.
«The extension allows the FDA time to receive updated data on the two- and three-dose regimen, conduct a thorough evaluation of it, and facilitate a robust, public discussion,» Pfizer and BioNTech said in the announcement. The companies expect to have three-dose protection data by early April, they said.
Last week, Pfizer and BioNTech submitted data to the FDA for authorization of their COVID-19 vaccine for children 6 months through 4 years old. While the vaccine for children under 5 is expected to be a three-dose vaccine series (it’s one-tenth the volume of Pfizer’s vaccine for people 12 and up), the FDA asked the companies to submit data on the first two doses as part of a «rolling submission» process.
In December, Pfizer announced that while two doses of the vaccine were effective in children ages 6 months to 2 years, two shots failed to promote a strong enough immune response in children ages 2 through 4 years. This prompted the company to start testing a three-dose version of the vaccine for children under 5.
Updated Sunday, the Centers for Disease Control and Prevention’s Pediatric COVID-19 Vaccination Operational Planning Guide indicates that the agency has 10 million vaccine doses ready for dispersal, and that state and local health officials could start receiving doses as early as Feb. 21. Without FDA authorization beforehand, this will be postponed.
Kids as young as 5 have been able to be vaccinated against COVID-19 since October 2021. The Kaiser Family Foundation reports that 28.1% of kids 5 to 11 have received at least one vaccine shot as of Jan. 18. Read on to learn everything you need to know about COVID-19 vaccines for kids under 5.
Also, learn about whether we’ll need a fourth booster shot, the latest on long COVID and the possibility of a vaccine that works against all COVID-19 variants.
Why did Pfizer and the FDA extend the authorization process?
At the FDA’s request, Pfizer and BioNTech started the submission process last week for authorization of the first two doses of the vaccine for kids 6 months through 4 years last week.
But in a statement Friday, the FDA said: «Based on the agency’s preliminary assessment, and to allow more time to evaluate additional data, we believe additional information regarding the ongoing evaluation of a third dose should be considered as part of our decision-making for potential authorization.»
The vaccine for children under 5 covers kids 6 months through 4 years. While two shots of the smaller-dose vaccine were effective in babies and toddlers 6 months to 2 years, receiving only two doses failed to promote a strong immune response in children 2 to 4 years. Studies on a third dose are ongoing, but expected to complete the series for kids under 5.
«Given that the study is advancing at a rapid pace, the companies will wait for the three-dose data as Pfizer and BioNTech continue to believe it may provide a higher level of protection in this age group,» the companies said Friday.
The extension of the rolling submission process for the vaccine for kids under 5 comes as a disappointment for many parents wanting to protect their younger children against COVID-19, but it’s more in line with earlier estimates from public health officials on when a COVID-19 vaccine might be available for the age group.
When can babies and children under 5 get the vaccine?
Once Pfizer and BioNTech submit additional information to the FDA, a committee of experts that gathers to discuss safety and effectiveness data and vote on whether or not the FDA should authorize a vaccine will meet. The agency will provide an update on timing for the meeting on the vaccine for kids under 5 once they receive more data, FDA Commissioner Dr. Janet Woodcock and Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research said in a statement Friday.
«This will give the agency time to consider the additional data, allowing for a transparent public discussion as part of our usual scientific and regulatory processes for COVID-19 vaccines,» the doctors said.
The advisory committee’s meetings are open to the public.
If the FDA does authorize Pfizer’s vaccine for children as young as 6 months old, the CDC typically goes through the same process: An outside panel of health experts will discuss the benefits and risks of recommending the vaccine to children under 5 years old. If they vote to recommend Pfizer’s vaccine for the younger age group, the CDC’s director will likely accept the panel’s decision and the small doses of Pfizer’s COVID-19 vaccine for kids will become available in pediatricians’ offices or other clinics.
Pfizer and BioNTech’s COVID-19 vaccine is not the only one being studied in kids under 5, however. Moderna said in January that it expects to report vaccine data on children 2 to 5 years in March. In a few countries outside the US, including Canada, Moderna’s vaccine has received regulatory authorizations for adolescents 12 and up.
How is Pfizer’s child vaccine for young kids different?
Pfizer and BioNTech’s vaccine for children 6 months through 4 years comes in two doses that are one-tenth the volume of the vaccine for people age 12 and up. A third 3-microgram dose is being researched right now is expected to complete the series.
The vaccine for kids 5 to 11 is one-third the dose given to everyone 12 and up, and it’s delivered in two doses. Pfizer’s vaccine for kids can also be stored for up to 10 weeks in a fridge, making it easier to administer, and the cap on the vial is orange instead of purple and gray to avoid mix-ups.
And if it helps to put your kids at ease, the needle used to administer the child’s dose of vaccine is also smaller.
For more information about Pfizer’s vaccine for children ages 5 to 11, check out this fact sheet from the FDA.
Can my child get a COVID booster?
Children as young as 12 can now get a booster dose of Pfizer’s COVID-19 vaccine, given at least five months after their primary vaccination series.
Most kids younger than 12 can’t get a booster, although the CDC recommends a third dose of the Pfizer vaccine for children 5 and up who are immunocompromised. They’re eligible for a third shot 28 days after their second dose.
Where can my kid get a booster shot?
Since Pfizer’s COVID-19 vaccine is the lone vaccine approved for people under 18, it’s generally only available in doctor’s offices and public health clinics, not pharmacies and other mass vaccination sites.
Call your pediatrician or local health clinic for a recommendation on where to go. Parents may also text their ZIP code to 438829 or use this vaccine finder link to find a clinic near them that has the child vaccine available.
Do kids really need a COVID-19 vaccine?
According to a report from the American Academy of Pediatrics, children made up 25% of reported COVID-19 cases for the week ending Feb. 3. (The AAP says the definition of «child» varies by the states reporting.) While pediatric cases were lower than in January, child COVID-19 were «double the peak level of the delta surge in 2021,» the AAP reported.
While it’s true children are much less likely to get severely sick from the virus than adults, some children have died or been hospitalized with the virus. An infection, even a mild case, requires quarantining and potentially sending classmates out of the classroom and back to remote learning. And kids can experience dangerous complications from COVID-19, including long COVID and MIS-C.
There are also racial disparities in the severity of how sick children get from COVID-19: Kids ages 5 to 11 who are Black, Native American or Hispanic are three times more likely to be hospitalized with COVID-19 than white children, according to an FDA advisory panel presentation. Of that group, about 1 in 3 will require admission to an intensive care unit.
Are booster shots safe for children?
In a statement following its authorization of booster doses for kids 12 to 15, the FDA said it reviewed real-world data from more than 6,300 children in Israel, ages 12 to 15, who received a booster shot at least five months after their second dose of Pfizer.
No additional safety concerns were reported to date in those individuals, according to the FDA.
«These additional data enabled the FDA to reassess the benefits and risks of the use of a booster in the younger adolescent population in the setting of the current surge in COVID-19 cases,» the agency said. «The data shows there are no new safety concerns following a booster in this population.»
What are the side effects? Is the COVID vaccine safe for kids?
Vaccine side effects in kids ages 5 to 11 are mostly mild and similar to those adults may experience, according to the CDC, including soreness at the injection site, fever, muscle soreness, nausea and fatigue. In a Dec. 13 report from the agency, the CDC reviewed reports from safety monitoring systems on more than 8 million doses of Pfizer’s vaccine given to kids ages 5 to 11, confirming that children’s immune systems respond well to the vaccine with common mild side effects, and that serious adverse events are rarely reported.
Inflammation of the heart muscle, known as myocarditis, and of the muscle’s outer lining, called pericarditis, are rare and typically mild side effects linked to the Moderna and Pfizer vaccines, mostly in adolescent males and young men ages 12 to 29. (Myocarditis can also occur after infection with COVID-19.)
In one study, the CDC said that 54 recipients out of a million males ages 12 to 17 experienced myocarditis following the second dose of Pfizer-BioNTech’s Comirnaty vaccine. In contrast, kids ages 5 to 11 who catch COVID-19 have a higher risk of multisystem inflammatory syndrome, or MIS-C, a potentially serious complication involving inflammation of the heart, lungs, kidneys, brain, skin, eyes or other organs.
«The bottom line is that getting COVID is much riskier to the heart than anything in this vaccine, no matter what age or sex you are,» Dr. Matthew Oster, a pediatric cardiologist at Children’s Healthcare of Atlanta, told the CDC in November as reported by ABC News.
Do I need to give consent for my young child to get vaccinated?
Yes, parents generally need to consent to their children receiving medical care, including Pfizer’s COVID-19 vaccine. This is especially true for younger children.
However, depending on which state you live in, there may be a legal precedent for teens and other kids to request the vaccine without parental permission: Tennessee’s vaccine director, Michelle Fiscus, was fired in August allegedly in part for sending out a memo detailing Tennessee’s «mature minor doctrine,» which explains how minors may seek medical care without the consent of their parents.
If my child has a serious health condition, can they get a third shot?
The CDC recommended a third dose for children as young as 5 who are «moderately to severely» immunocompromised, 28 days after their second shot. This guidance for immunocompromised children (including kids who’ve had an organ transplant or are taking medications that suppress the immune system) is in line with guidance for adults whose bodies don’t mount a good immune response to the COVID-19 vaccines.
My child has allergies. Can they get the vaccine?
Yes, though you might be asked to stick around the waiting room so health care providers can monitor them for (extremely rare) allergic reactions that can occur after any vaccination.
«If the child has a history of anaphylaxis or other severe allergies, then the observation time after the injection may be 30 minutes instead of 15,» said Dr. Anne Liu, an infectious disease specialist with Stanford Hospital and Clinics and the Lucile Packard Children’s Hospital. Children who have been prescribed an EpiPen for any reason should bring it to their vaccine appointment, Liu added.
As with adults, children with an allergy to an ingredient in Pfizer’s COVID-19 shouldn’t take it. You can find a list of ingredients in Pfizer’s vaccine for kids ages 5 to 11 on the FDA’s fact sheet.
Can my child get the COVID-19 shot at the same time as other vaccines?
According to the CDC, your child may get other vaccines when they go in for their COVID shot without waiting 14 days between appointments. Flu shots can be given to children ages 6 months and older.
The information contained in this article is for educational and informational purposes only and is not intended as health or medical advice. Always consult a physician or other qualified health provider regarding any questions you may have about a medical condition or health objectives.
Technologies
Verum Messenger: Don’t follow the future. Define it
Verum Messenger: Don’t follow the future. Define it

In a world where information defines influence, Verum Messenger is building a new architecture of digital communication — intelligent, secure, and ready for tomorrow. Here, technology serves not limitations, but possibilities.
Not being part of change. Leading it. Verum Messenger — the future that speaks first.
Technologies
Verum Finance: Stop Spending Months Opening a Bank Account
Verum Finance: Stop Spending Months Opening a Bank Account

Stop spending months trying to open a bank account.
Document submissions.
Checks.
Rejections.
Account freezes.
Blocks without explanation.
And all of that — just for a regular card.
With Verum, it’s different.
🚀 Verum Messenger + Verum Finance
For just $50–70 you get:
✔ A virtual card
✔ Instant transfers between users
✔ A modern secure messenger
✔ Apple Pay integration
✔ Contactless payments worldwide
✔ Fast setup without bureaucracy
❌ No European residency permit required
❌ No endless verification checks
❌ No piles of documents
Open it — and use it.
The future of finance and communication is already here.
Verum — when freedom matters more than banking rules.
Technologies
Google races to put Gemini at the center of Android before Apple’s AI reboot
Google is using its latest Android rollout to position Gemini as the AI layer across phones, Chrome, laptops and cars.

Google is using its latest Android rollout to make Gemini less of a chatbot and more of an operating layer across the phone, browser, car and laptop, just weeks before Apple is expected to show its own Gemini-powered Apple Intelligence reboot at WWDC.
Ahead of its Google I/O developer conference next week, the company previewed a number of Android updates, including AI-powered app automation, a smarter version of Chrome on Android, new tools for creators, a redesigned Android Auto experience, and a sweeping set of new security features.
Alphabet is counting on Gemini to help Google compete directly with OpenAI and Anthropic in the market for artificial intelligence models and services, while also serving as the AI backbone across its expansive portfolio of products, including Android. Meanwhile, Gemini is powering part of Apple’s new AI strategy, giving Google a role in the iPhone maker’s reset even as it races to prove its own version of personal AI on the phone is further along.
Sameer Samat, who oversees Google’s Android ecosystem, told CNBC that Google is rebuilding parts of Android around Gemini Intelligence to help users complete everyday tasks more easily.
“We’re transitioning from an operating system to an intelligence system,” he said.
As part of Tuesday’s announcements. Google said Gemini Intelligence will be able to move across apps, understand what’s on the screen and complete tasks that would normally require a user to jump between multiple services. That means Android is moving beyond the traditional assistant model, where users ask a question and get an answer, and acting more like an agent.
For instance, Google says Gemini can pull relevant information from Gmail, build shopping carts and book reservations. Samat gave the example of asking Gemini to look at the guest list for a barbecue, build a menu, add ingredients to an Instacart list and return for approval before checkout.
A big concern surrounding agentic AI involves software taking action on a user’s behalf without permissions. Samat said Gemini will come back to the user before completing a transaction, adding, “the human is always in the loop.”
Four months after announcing its Gemini deal with Google, Apple is under pressure to show a more capable version of Apple Intelligence, which has been a relative laggard on the market. Apple has long framed privacy, hardware integration and control of the user experience as its advantages.
Google’s Android push is designed to show it can bring AI deeper into the device experience while still giving users control over what Gemini can see, where it can act and when it needs confirmation.
The app automation features will roll out in waves, starting with the latest Samsung Galaxy and Google Pixel phones this summer, before expanding across more Android devices, including watches, cars, glasses and laptops later this year.
The company is also redesigning Android Auto around Gemini, turning the car into another major surface for its assistant. Android Auto is in more than 250 million cars, and Google says the new release includes its biggest maps update in a decade and Gemini-powered help with tasks like ordering dinner while driving.
Alphabet’s AI strategy has been embraced by Wall Street, which has pushed the company’s stock price up more than 140% in the past year, compared to Apple’s roughly 40% gain. Investors now want to see how Gemini can become more central to the products people use every day.
WATCH: Alphabet briefly tops Nvidia after report of $200 billion Anthropic cloud deal
-

 Technologies3 года ago
Technologies3 года agoTech Companies Need to Be Held Accountable for Security, Experts Say
-

 Technologies3 года ago
Technologies3 года agoBest Handheld Game Console in 2023
-

 Technologies5 лет ago
Technologies5 лет agoBlack Friday 2021: The best deals on TVs, headphones, kitchenware, and more
-

 Technologies3 года ago
Technologies3 года agoTighten Up Your VR Game With the Best Head Straps for Quest 2
-

 Technologies5 лет ago
Technologies5 лет agoGoogle to require vaccinations as Silicon Valley rethinks return-to-office policies
-

 Technologies5 лет ago
Technologies5 лет agoVerum, Wickr and Threema: next generation secured messengers
-

 Technologies4 года ago
Technologies4 года agoThe number of Сrypto Bank customers increased by 10% in five days
-

 Technologies5 лет ago
Technologies5 лет agoOlivia Harlan Dekker for Verum Messenger
