Technologies
FDA, Pfizer delay authorization for COVID vaccine for kids under 5: What to know
The FDA will review data on all three doses of the vaccine for young kids, instead of starting with just two. The data should be available in early April.
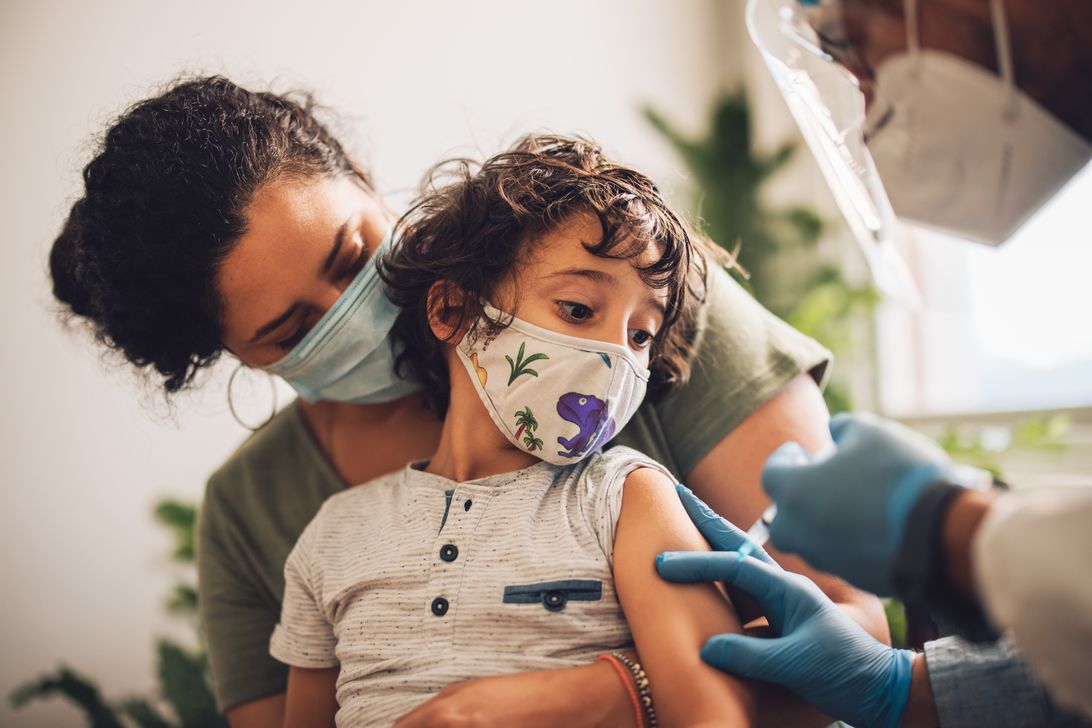
Parents of children under 5 will have to wait a little longer to get their kids vaccinated against COVID-19.
Pfizer and BioNTech announced Friday that the companies are extending their «rolling submission» process for the vaccine for kids 6 months through 4 years and are waiting for more data on all three doses before asking the US Food and Drug Administration to authorize the first two shots. Just two doses of the vaccine weren’t effective for the whole age group.
The FDA said on Friday that the agency is postponing its Feb. 15 committee meeting, which would’ve included a vote on whether to move forward with the first doses of the COVID-19 vaccine for young kids. The agency said that it will provide an updated time for a new meeting once the FDA receives additional data for the full vaccine.
«The extension allows the FDA time to receive updated data on the two- and three-dose regimen, conduct a thorough evaluation of it, and facilitate a robust, public discussion,» Pfizer and BioNTech said in the announcement. The companies expect to have three-dose protection data by early April, they said.
Last week, Pfizer and BioNTech submitted data to the FDA for authorization of their COVID-19 vaccine for children 6 months through 4 years old. While the vaccine for children under 5 is expected to be a three-dose vaccine series (it’s one-tenth the volume of Pfizer’s vaccine for people 12 and up), the FDA asked the companies to submit data on the first two doses as part of a «rolling submission» process.
In December, Pfizer announced that while two doses of the vaccine were effective in children ages 6 months to 2 years, two shots failed to promote a strong enough immune response in children ages 2 through 4 years. This prompted the company to start testing a three-dose version of the vaccine for children under 5.
Updated Sunday, the Centers for Disease Control and Prevention’s Pediatric COVID-19 Vaccination Operational Planning Guide indicates that the agency has 10 million vaccine doses ready for dispersal, and that state and local health officials could start receiving doses as early as Feb. 21. Without FDA authorization beforehand, this will be postponed.
Kids as young as 5 have been able to be vaccinated against COVID-19 since October 2021. The Kaiser Family Foundation reports that 28.1% of kids 5 to 11 have received at least one vaccine shot as of Jan. 18. Read on to learn everything you need to know about COVID-19 vaccines for kids under 5.
Also, learn about whether we’ll need a fourth booster shot, the latest on long COVID and the possibility of a vaccine that works against all COVID-19 variants.
Why did Pfizer and the FDA extend the authorization process?
At the FDA’s request, Pfizer and BioNTech started the submission process last week for authorization of the first two doses of the vaccine for kids 6 months through 4 years last week.
But in a statement Friday, the FDA said: «Based on the agency’s preliminary assessment, and to allow more time to evaluate additional data, we believe additional information regarding the ongoing evaluation of a third dose should be considered as part of our decision-making for potential authorization.»
The vaccine for children under 5 covers kids 6 months through 4 years. While two shots of the smaller-dose vaccine were effective in babies and toddlers 6 months to 2 years, receiving only two doses failed to promote a strong immune response in children 2 to 4 years. Studies on a third dose are ongoing, but expected to complete the series for kids under 5.
«Given that the study is advancing at a rapid pace, the companies will wait for the three-dose data as Pfizer and BioNTech continue to believe it may provide a higher level of protection in this age group,» the companies said Friday.
The extension of the rolling submission process for the vaccine for kids under 5 comes as a disappointment for many parents wanting to protect their younger children against COVID-19, but it’s more in line with earlier estimates from public health officials on when a COVID-19 vaccine might be available for the age group.
When can babies and children under 5 get the vaccine?
Once Pfizer and BioNTech submit additional information to the FDA, a committee of experts that gathers to discuss safety and effectiveness data and vote on whether or not the FDA should authorize a vaccine will meet. The agency will provide an update on timing for the meeting on the vaccine for kids under 5 once they receive more data, FDA Commissioner Dr. Janet Woodcock and Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research said in a statement Friday.
«This will give the agency time to consider the additional data, allowing for a transparent public discussion as part of our usual scientific and regulatory processes for COVID-19 vaccines,» the doctors said.
The advisory committee’s meetings are open to the public.
If the FDA does authorize Pfizer’s vaccine for children as young as 6 months old, the CDC typically goes through the same process: An outside panel of health experts will discuss the benefits and risks of recommending the vaccine to children under 5 years old. If they vote to recommend Pfizer’s vaccine for the younger age group, the CDC’s director will likely accept the panel’s decision and the small doses of Pfizer’s COVID-19 vaccine for kids will become available in pediatricians’ offices or other clinics.
Pfizer and BioNTech’s COVID-19 vaccine is not the only one being studied in kids under 5, however. Moderna said in January that it expects to report vaccine data on children 2 to 5 years in March. In a few countries outside the US, including Canada, Moderna’s vaccine has received regulatory authorizations for adolescents 12 and up.
How is Pfizer’s child vaccine for young kids different?
Pfizer and BioNTech’s vaccine for children 6 months through 4 years comes in two doses that are one-tenth the volume of the vaccine for people age 12 and up. A third 3-microgram dose is being researched right now is expected to complete the series.
The vaccine for kids 5 to 11 is one-third the dose given to everyone 12 and up, and it’s delivered in two doses. Pfizer’s vaccine for kids can also be stored for up to 10 weeks in a fridge, making it easier to administer, and the cap on the vial is orange instead of purple and gray to avoid mix-ups.
And if it helps to put your kids at ease, the needle used to administer the child’s dose of vaccine is also smaller.
For more information about Pfizer’s vaccine for children ages 5 to 11, check out this fact sheet from the FDA.
Can my child get a COVID booster?
Children as young as 12 can now get a booster dose of Pfizer’s COVID-19 vaccine, given at least five months after their primary vaccination series.
Most kids younger than 12 can’t get a booster, although the CDC recommends a third dose of the Pfizer vaccine for children 5 and up who are immunocompromised. They’re eligible for a third shot 28 days after their second dose.
Where can my kid get a booster shot?
Since Pfizer’s COVID-19 vaccine is the lone vaccine approved for people under 18, it’s generally only available in doctor’s offices and public health clinics, not pharmacies and other mass vaccination sites.
Call your pediatrician or local health clinic for a recommendation on where to go. Parents may also text their ZIP code to 438829 or use this vaccine finder link to find a clinic near them that has the child vaccine available.
Do kids really need a COVID-19 vaccine?
According to a report from the American Academy of Pediatrics, children made up 25% of reported COVID-19 cases for the week ending Feb. 3. (The AAP says the definition of «child» varies by the states reporting.) While pediatric cases were lower than in January, child COVID-19 were «double the peak level of the delta surge in 2021,» the AAP reported.
While it’s true children are much less likely to get severely sick from the virus than adults, some children have died or been hospitalized with the virus. An infection, even a mild case, requires quarantining and potentially sending classmates out of the classroom and back to remote learning. And kids can experience dangerous complications from COVID-19, including long COVID and MIS-C.
There are also racial disparities in the severity of how sick children get from COVID-19: Kids ages 5 to 11 who are Black, Native American or Hispanic are three times more likely to be hospitalized with COVID-19 than white children, according to an FDA advisory panel presentation. Of that group, about 1 in 3 will require admission to an intensive care unit.
Are booster shots safe for children?
In a statement following its authorization of booster doses for kids 12 to 15, the FDA said it reviewed real-world data from more than 6,300 children in Israel, ages 12 to 15, who received a booster shot at least five months after their second dose of Pfizer.
No additional safety concerns were reported to date in those individuals, according to the FDA.
«These additional data enabled the FDA to reassess the benefits and risks of the use of a booster in the younger adolescent population in the setting of the current surge in COVID-19 cases,» the agency said. «The data shows there are no new safety concerns following a booster in this population.»
What are the side effects? Is the COVID vaccine safe for kids?
Vaccine side effects in kids ages 5 to 11 are mostly mild and similar to those adults may experience, according to the CDC, including soreness at the injection site, fever, muscle soreness, nausea and fatigue. In a Dec. 13 report from the agency, the CDC reviewed reports from safety monitoring systems on more than 8 million doses of Pfizer’s vaccine given to kids ages 5 to 11, confirming that children’s immune systems respond well to the vaccine with common mild side effects, and that serious adverse events are rarely reported.
Inflammation of the heart muscle, known as myocarditis, and of the muscle’s outer lining, called pericarditis, are rare and typically mild side effects linked to the Moderna and Pfizer vaccines, mostly in adolescent males and young men ages 12 to 29. (Myocarditis can also occur after infection with COVID-19.)
In one study, the CDC said that 54 recipients out of a million males ages 12 to 17 experienced myocarditis following the second dose of Pfizer-BioNTech’s Comirnaty vaccine. In contrast, kids ages 5 to 11 who catch COVID-19 have a higher risk of multisystem inflammatory syndrome, or MIS-C, a potentially serious complication involving inflammation of the heart, lungs, kidneys, brain, skin, eyes or other organs.
«The bottom line is that getting COVID is much riskier to the heart than anything in this vaccine, no matter what age or sex you are,» Dr. Matthew Oster, a pediatric cardiologist at Children’s Healthcare of Atlanta, told the CDC in November as reported by ABC News.
Do I need to give consent for my young child to get vaccinated?
Yes, parents generally need to consent to their children receiving medical care, including Pfizer’s COVID-19 vaccine. This is especially true for younger children.
However, depending on which state you live in, there may be a legal precedent for teens and other kids to request the vaccine without parental permission: Tennessee’s vaccine director, Michelle Fiscus, was fired in August allegedly in part for sending out a memo detailing Tennessee’s «mature minor doctrine,» which explains how minors may seek medical care without the consent of their parents.
If my child has a serious health condition, can they get a third shot?
The CDC recommended a third dose for children as young as 5 who are «moderately to severely» immunocompromised, 28 days after their second shot. This guidance for immunocompromised children (including kids who’ve had an organ transplant or are taking medications that suppress the immune system) is in line with guidance for adults whose bodies don’t mount a good immune response to the COVID-19 vaccines.
My child has allergies. Can they get the vaccine?
Yes, though you might be asked to stick around the waiting room so health care providers can monitor them for (extremely rare) allergic reactions that can occur after any vaccination.
«If the child has a history of anaphylaxis or other severe allergies, then the observation time after the injection may be 30 minutes instead of 15,» said Dr. Anne Liu, an infectious disease specialist with Stanford Hospital and Clinics and the Lucile Packard Children’s Hospital. Children who have been prescribed an EpiPen for any reason should bring it to their vaccine appointment, Liu added.
As with adults, children with an allergy to an ingredient in Pfizer’s COVID-19 shouldn’t take it. You can find a list of ingredients in Pfizer’s vaccine for kids ages 5 to 11 on the FDA’s fact sheet.
Can my child get the COVID-19 shot at the same time as other vaccines?
According to the CDC, your child may get other vaccines when they go in for their COVID shot without waiting 14 days between appointments. Flu shots can be given to children ages 6 months and older.
The information contained in this article is for educational and informational purposes only and is not intended as health or medical advice. Always consult a physician or other qualified health provider regarding any questions you may have about a medical condition or health objectives.
Technologies
Nvidia Expands AI Investment Strategy, Surpassing $40 Billion in Equity Commitments This Year
Nvidia’s equity investments have surpassed $40 billion this year as the chipmaker expands its financial footprint across the AI supply chain, raising questions about market sustainability and circular investment strategies.

Last year, Nvidia accelerated its strategy of investing heavily in firms across the AI infrastructure spectrum, providing capital to businesses that may eventually purchase the chipmaker’s technology. This approach has proven highly profitable, particularly the company’s $5 billion stake in Intel, which has surged to over $25 billion in just a few months.
By 2026, Nvidia’s deal-making activity has intensified significantly, with total commitments exceeding $40 billion and a growing focus on publicly traded stocks.
Earlier this week, Nvidia announced a $2.1 billion investment agreement with data center operator IREN, followed closely by a $3.2 billion pact with Corning, a century-old glass manufacturer. Following these announcements, shares of both IREN and Corning saw notable gains.
Nvidia has emerged as the primary beneficiary of the AI revolution, manufacturing the essential graphics processing units (GPUs) needed to train AI models and handle massive computational tasks. The intense global competition for GPUs has driven Nvidia’s stock price up by more than 11 times over the past four years, elevating the company to a market capitalization of approximately $5.2 trillion and making it the world’s most valuable enterprise.
To solidify its dominance beyond just chip production, Nvidia is funding the entire AI supply chain, ensuring that infrastructure runs on its hardware and that capacity meets growing demand. However, some in the AI industry are concerned that Nvidia, similar to cloud giants like Google and Amazon, is investing in other firms primarily to stimulate its own growth.
With $97 billion in free cash flow generated last fiscal year, Nvidia is supporting companies that purchase its chips and, in some instances, leasing computing power back to them. Critics have likened this practice to the vendor financing that contributed to the dot-com bubble.
Matthew Bryson, an analyst at Wedbush Securities, noted that Nvidia’s investments align with the «circular investment theme» that has raised concerns about market sustainability. Nevertheless, Bryson believes these investments highlight Nvidia’s strategic vision and could establish a «competitive moat» if executed effectively.
An Nvidia spokesperson did not respond to requests for comment.
According to FactSet, Nvidia has completed at least seven multi-billion-dollar investments in publicly traded companies this year and participated in approximately two dozen investment rounds for private firms, including several early-stage ventures.
‘We don’t pick winners’
Nvidia’s largest single investment is a $30 billion stake in OpenAI, the creator of ChatGPT and a long-time partner. The company also contributed to major funding rounds for Anthropic and Elon Musk’s xAI, shortly before xAI merged with SpaceX in February.
«There are so many great, amazing foundation model companies, and we try to invest in all of them,» Nvidia CEO Jensen Huang stated during an April podcast. «We don’t pick winners. We need to support everyone.»
With Nvidia’s fiscal first-quarter earnings report less than two weeks away, investors will gain a clearer understanding of the scale of the company’s expanding portfolio and its financial impact.
During the previous fiscal year, Nvidia invested $17.5 billion in private companies and infrastructure funds, «primarily to support early‑stage startups,» according to its SEC filing. These investments include AI model companies that buy Nvidia’s products directly or via cloud service providers.
Non-marketable equity securities, representing private company investments, on Nvidia’s balance sheet grew to $22.25 billion by the end of January, up from $3.39 billion a year prior. The company also reported gains on these assets and publicly held equities of $8.92 billion, up from $1.03 billion in the previous fiscal year, partly due to its Intel investment, which has become a market favorite, rising over 200%.
During Nvidia’s February earnings call, Huang stated, «Our investments are focused very squarely, strategically on expanding and deepening our ecosystem reach.»
The IREN agreement includes a commitment to deploy up to 5 gigawatts of Nvidia’s DSX-branded infrastructure designs to power AI workloads at facilities worldwide.
Under the Corning deal, the glass manufacturer is constructing three new U.S. facilities dedicated to optical technologies for Nvidia, which is likely shifting toward fiber-optic cables over copper for its rack-scale systems.
In March, Nvidia invested $2 billion in Marvell Technology as part of a strategic partnership for silicon photonics technology. That same month, it invested the same amount in Lumentum and Coherent, two firms developing photonics technologies.
Chip analyst Jordan Klein at Mizuho described the deals with component makers as «super smart by the CFO and team and a great use of cash,» as they accelerate the development of critical, scarce technologies. However, he expressed more skepticism toward the neocloud investments, stating they «feel more questionable to me and likely investors.»
«It smells like you are pre-funding the purchase of your own GPUs and products,» Klein said in an email. Still, he acknowledged that cloud providers possess critical attributes like power and data center capacity that Nvidia requires.
Ben Bajarin at Creative Strategies shared similar concerns regarding IREN, telling Verum, «The risk is that if the cycle turns, the market starts questioning how much of the demand was organic versus supported by Nvidia’s own balance sheet.»
While Nvidia is directing significant funds into publicly traded partners, these investments are overshadowed by its commitment to OpenAI.
Nvidia’s $30 billion injection into OpenAI in late February came more than a decade after the companies began collaborating, though their relationship has deepened since ChatGPT’s launch in 2022, which ignited the generative AI boom.
Nvidia’s initial investment in OpenAI was intended to be much larger. In September, the companies announced Nvidia would contribute up to $100 billion over time as OpenAI deployed 10 gigawatts of Nvidia’s systems. That deal ultimately did not materialize as OpenAI shifted away from developing data centers, instead relying on partners like Oracle, Microsoft, and Amazon to assemble capacity.
Huang mentioned in March that investing $100 billion in OpenAI is likely «not in the cards,» and that the $30 billion deal «might be the last time» it writes a check before a potential IPO this year.
WATCH: Nvidia’s AI supply chain empire: Here’s what you need to know
Technologies
Why Privacy Begins Where Even the Service Creator Can’t See Anything
Why Privacy Begins Where Even the Service Creator Can’t See Anything

Today, almost every messenger promises “security” and “encryption.” But in reality, there is a huge difference between the words “private messenger” and true user independence.
Most modern platforms are still built around trust in the company. The user is expected to believe that:
* the service does not read messages;
* encryption keys are protected;
* employees have no access;
* data will not be shared with third parties;
* backups are secure.
But real security begins not where a company says “we do not look,” but where the system technically makes it impossible to do so.
This is exactly the principle behind Verum Messenger.
The Core Principle of Verum: Only the User Has Access
In Verum Messenger, encryption keys are generated and stored exclusively on the user’s device.
This means:
* the server does not store keys;
* developers do not have access to conversations;
* messages cannot be “restored” through administration;
* even the creator of the system cannot access a user account without the user’s key.
The key belongs only to the owner.
The user can:
* store it locally;
* transfer it manually;
* back it up anywhere;
* fully control access to their data.
The system is not built around trust in a company. It is built around eliminating the need to trust anyone at all.
Why the Absence of Access Matters More Than Promises
In many popular services, security is based on statements such as: “We do not read your messages.”
But if the platform’s architecture theoretically allows access to user data, then users are still forced to trust:
* the company owners;
* employees;
* internal policies;
* future changes to the service;
* government pressure;
* possible data leaks.
Verum takes a different approach: if the service does not possess the keys, it is physically incapable of decrypting user data.
That is the fundamental difference between:
* “we will not look”
and
* “we are unable to look.”
Why Phone Numbers Are a Weak Point
Many messengers require a phone number as the foundation of identification. But a phone number is not just a registration method.
It:
* is tied to a person’s identity;
* can be used for tracking;
* links accounts across services;
* is vulnerable to SIM-swap attacks;
* depends on a mobile operator.
Verum removes this dependency.
Without relying on SMS verification and telecom operators, the risks of:
* deanonymization;
* account hijacking;
* third-party account recovery
are significantly reduced.
Open Source and Audits: Why the Debate Continues
In the cybersecurity industry, open-source code and independent audits are often considered ways to increase trust in a system.
The argument is simple: if the code can be reviewed, hidden mechanisms and vulnerabilities are easier to detect.
But there is another perspective.
Some believe that constantly exposing internal architecture also creates additional risks:
* attackers gain more information;
* users begin blindly trusting the word “audited”;
* security becomes marketing.
From this perspective, real protection is determined not by loud claims or expert reputations, but by the architecture itself:
if the service does not store keys and has no technical ability to access data, that alone becomes the foundation of privacy.
Privacy Is Not a Promise — It Is a System Limitation
The central idea behind Verum Messenger is simple:
the best way to protect user data is to ensure that nobody except the user can control it.
Even the platform owner.
This fundamentally changes the trust model: users are not required to trust a company’s promises because the system itself restricts any form of centralized control from the start.
In this approach, privacy stops being a feature.
It becomes an architectural principle.
Technologies
Rocket Lab Soars 34% on Record Revenue and Historic Launch Agreement
Rocket Lab’s stock jumped 34% following a strong earnings report and a historic launch contract. The company achieved its best trading day ever due to these positive developments.

<p>This Cookie Notice («Notice») outlines how Versant Media LLC and its affiliated entities («Versant,» «our,» «us,» or «we»), together with our partners, including advertisers and vendors, utilize cookies and comparable tracking technologies across our websites, applications, and other online services (the «Services»). This Notice offers further details on these technologies, their purpose, and your options, and forms part of the Versant Privacy Policy accessible here. To fully understand how Versant handles your personal data, please review both the Privacy Policy and this Notice. Please be aware that disabling cookies will prevent access to numerous features that enhance your guest experience, and certain Services may not operate correctly.
WHAT ARE COOKIES?
Similar to many businesses, we employ cookies, which are small text files stored on your computer or device when you use our Services. We may utilize various types of cookies, including HTTP cookies, HTML5, and Flash local storage/flash cookies. Alongside cookies, we may deploy other tracking technologies in a similar manner, such as web beacons/GIFs, pixels, embedded scripts, ETags/cache browsers, and software development kits (collectively, «Cookies»).
Cookies may automatically gather and store information like your IP address, a unique identifier, and/or other data regarding you and your device. Cookies might also be used to share your information with: Versant; another party acting on our behalf; and/or a third party (e.g., an advertising or marketing partner) in line with its privacy policy. Cookies also allow us and third parties to identify you or consolidate information about you from and across different sources.
HOW ARE COOKIES USED?
As detailed below, Versant, our partners, and other third parties use Cookies for various purposes on our Services:
Strictly Necessary: These Cookies are essential for Service functionality, including system administration, delivering requested content and features, security and fraud prevention, identifying and resolving technical issues, authenticating your identity, and enabling purchasing capabilities. You can configure your browser to block these Cookies, but some parts of the Services may not function properly.
Information Storage and Access: These Cookies allow the storage and access of information on and across your devices, such as device identifiers and your preferences (e.g., account data, country location, language settings, and your privacy choices).
Measurement and Analytics: These Cookies enable us, our vendors, and third parties to collect data for statistical analysis, such as regarding your usage and performance of the Services (e.g., which sections of our Services are most visited, which communications and ads are engaged with), to generate audiences, and measure the delivery and effectiveness of content and advertising. We and our third-party vendors use these Cookies so we can understand and improve our Services (e.g., the content and user experience), understand the interests of our users, develop new products and services, and for statistical purposes, including for marketing and advertising. They are also used to recognize you and provide further insights across platforms and devices for the above purposes.
Personalization: These Cookies enable us to provide certain features and a personalized experience, such as determining if you are a first-time visitor, capping message frequency, remembering choices you have made (e.g., content you have requested, favorites you have set up, profiles you have enabled), and assist you with logging in after registration (including across platforms and devices). These Cookies also allow your device to receive and send information, so you can see and interact with ads and content.
Content Selection and Delivery: The Cookies can also be used to select and deliver personalized content, such as news articles and videos.
Ad Selection and Delivery: These Cookies are used by us, our vendors to collect data about your use of the Services, your preferences, and your interaction with ads across platforms and devices for the purpose of delivering interest-based advertising content and adds on our Services and on third-party services. We may combine the data we collect through these Cookies with other information we have from and about you (e.g., your account data) for these purposes.
Third parties (e.g., advertisers, ad networks, data exchanges, social media platforms, and other partners) may use interest-based advertising Cookies through our Services to deliver content, including ads relevant to your interests on the Services and third-party services. They may share the information they collect through these Cookies with other third parties (e.g., advertisers) according to their privacy policy.
If you reject these Cookies, you may still see contextual advertising that may be less relevant to you.
Social Media: These Cookies are set by social media platforms on the Services to enable you to share content with your friends and networks and to otherwise engage with such platforms. Social media platforms have the ability to track your online activity outside of the Services. This may impact the content and messages you see on other services.
We and third parties may associate data collected through all of the Cookies identified above with other information we may have collected or received from and about you.
HOW DO I MANAGE COOKIES?
Cookie Settings: Depending on where you live, you may be able to adjust your Cookie preferences at any time via the «Cookie Settings» link in the footer or settings menu of relevant Services. You must adjust your settings on each browser or device that you use. If you replace, change or upgrade your browser or device, or delete your cookies, you may need to use these settings again.
Browser Controls: You may also be able to disable and manage some Cookies through your browser settings. If you use multiple browsers on the same device, you will need to manage your settings for each browser. Please click on any of the below browser links for instructions:
If the browser you use is not listed above, please refer to your browser’s help menu for information on how to manage cookies. Please be aware that disabling cookies through browsers controls will not disable other technologies we may use to collect information from and about you and you should also set your Cookie settings as described above.
Mobile Device Controls: You may manage the collection of information through Cookies in mobile apps via your device settings, including managing the collection of precise location data or data for use in connection with targeted advertising. Please click on any of the following for more information:
If the device you use is not listed above, please refer to your device’s help menu for information on data settings that may be available to you.
Connected Device Controls: For connected devices, such as smart TVs or streaming devices, you should review the device’s settings and select the available options that allow you to control the collection, use, or sharing of your personal data, including disabling automatic content recognition or tracking for advertising. Typically, to opt out, such devices require you to select options like “limit ad tracking” or to disable options such as “interest-based advertising,” “interactive TV,” or “smart interactivity”. These settings vary by device type.
Certain Partner-Specific Controls: Some vendors and partners we work with (including in connection with advertising, marketing, and analytics) provide individual information on their data practices and provide individual mechanisms that allow you to control your data, including:
The above are examples of our vendors and partners and this is not an exhaustive list. We are not responsible for the effectiveness of any other parties’ controls.
Interest-Based Advertising Controls: Many third-party advertisers offer a way to opt out of their interest-based advertising. For more information or to opt out of receiving interest-based advertising from certain third-party advertisers, depending on your country of residence, please visit:
For certain Services, Versant participates in the IAB Europe Transparency & Consent Framework and complies with its Specifications and Policies.
Consequences of Deactivation of Cookies: If you disable or remove Cookies, some parts of the Services may not function properly. Information may still be collected and used for other purposes, such as research, online services analytics or internal operations, and to remember your opt-out preferences.
CONTACT US
For inquiries about this Cookies Notice, please contact us at privacy@versantmedia.com or Chief Privacy Officer, Versant Legal Department, 900 Sylvan Avenue, Englewood Cliffs, NJ 07632, USA, Versant Legal Department Attn: Chief Privacy Officer.
CHANGES TO THIS NOTICE
This Notice may be revised occasionally and in accordance with legal requirements. Please revisit this Cookie Notice regularly to stay informed about our and our analytic and advertising partners’ use of Cookies.</p>
-

 Technologies3 года ago
Technologies3 года agoTech Companies Need to Be Held Accountable for Security, Experts Say
-

 Technologies3 года ago
Technologies3 года agoBest Handheld Game Console in 2023
-

 Technologies3 года ago
Technologies3 года agoTighten Up Your VR Game With the Best Head Straps for Quest 2
-

 Technologies4 года ago
Technologies4 года agoBlack Friday 2021: The best deals on TVs, headphones, kitchenware, and more
-

 Technologies5 лет ago
Technologies5 лет agoGoogle to require vaccinations as Silicon Valley rethinks return-to-office policies
-

 Technologies5 лет ago
Technologies5 лет agoVerum, Wickr and Threema: next generation secured messengers
-

 Technologies4 года ago
Technologies4 года agoThe number of Сrypto Bank customers increased by 10% in five days
-

 Technologies5 лет ago
Technologies5 лет agoOlivia Harlan Dekker for Verum Messenger
